Overview:
Set up under the aegis of the Office of Principal Scientific Adviser (PSA) to the Government of India, The Empowered Committee for Animal Health (ECAH) is chaired by the Principal Scientific Adviser (PSA), Government of India and vice-chaired by the Secretary, Department of Animal Husbandry and Dairying (DAHD). This Committee brings together various animal health experts to collaborate on achieving the completion of its goals. The members represented on the committee include experts from Central Drug Standard Control Organization (CDSCO), Department of Biotechnology (DBT), Indian Council of Agricultural Research (ICAR), Indian Council of Medical Research (ICMR), Indian Veterinary Research Institute (IVRI), Veterinary Council of India (VCI), and prominent academics from notable veterinary institutes and industry.
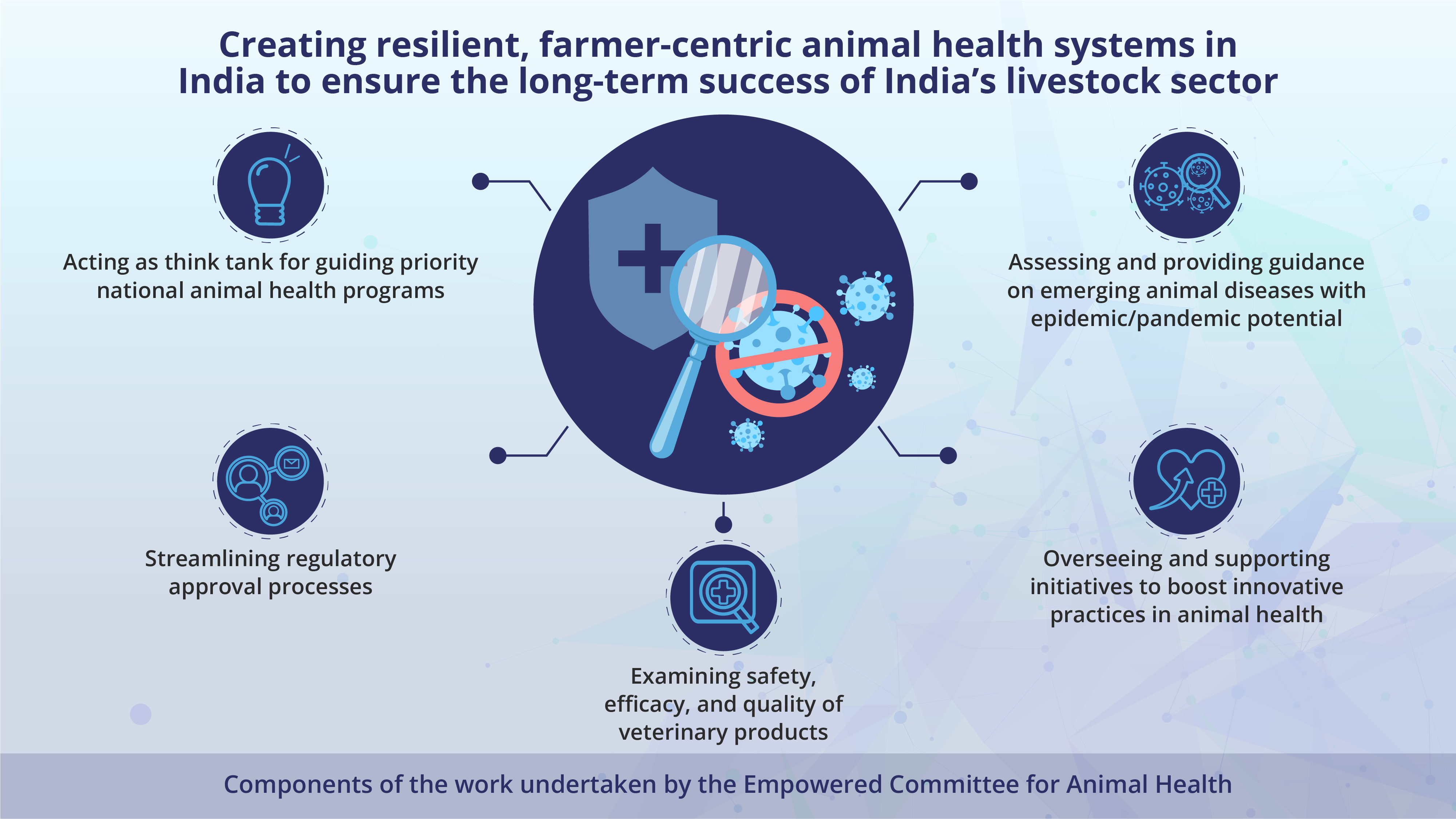
ECAH is focused on creating resilient, farmer-centric animal health systems in India and bring forth changes to ensure the long-term success of India’s livestock sector by -
● Acting as a think tank to provide inputs on animal health programmes of national importance
● Streamlining and improving the process of regulatory approval to protect public, animal health, and the environment by examining the safety, efficacy, and quality of veterinary products
● Overseeing and supporting programmes/initiatives to drive uptake of innovative practices in the Indian animal health sector
● Assessing the threat of and offering guidance on emerging animal diseases that have epidemic/pandemic potential
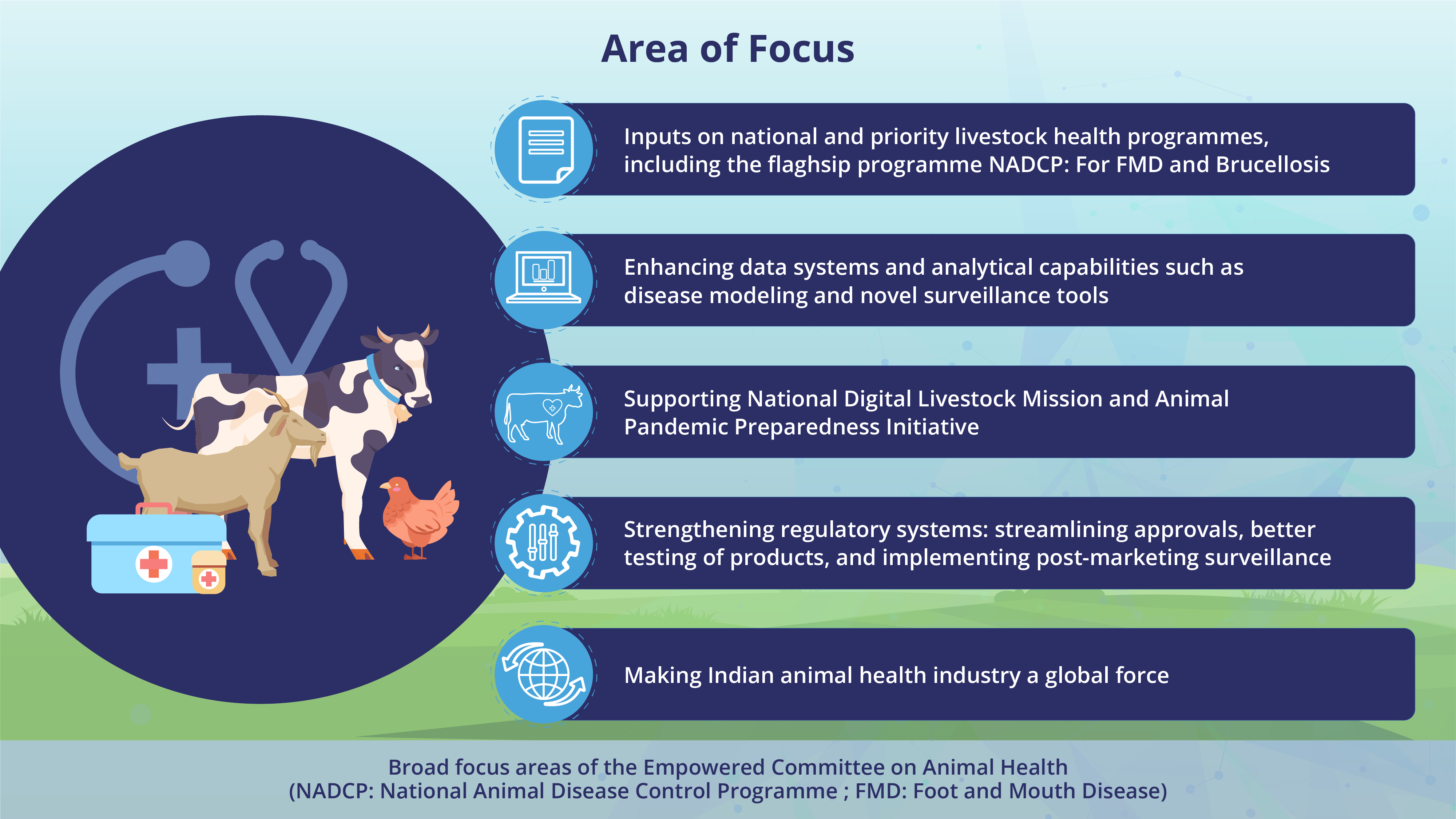
Areas of focus:
● Providing inputs on priority programmes in livestock health such NADCP: For FMD and Brucellosis - flagship program, One Health Support Unit (OHSU), and other programmes of national importance.
● Enhancing data systems and analytic capabilities to better inform decision-making in the sector.
● Supporting National Digital Livestock Mission (NDLM) and Animal Pandemic Preparedness Initiative.
● Strengthening the regulatory systems through streamlined approval processes, enhanced testing of the products and implementing post-marketing surveillance to ensure quality products reach the market.
● Supporting and enabling the animal health industry to be a global force.